Protect your family from respiratory illnesses. Schedule your immunization here >
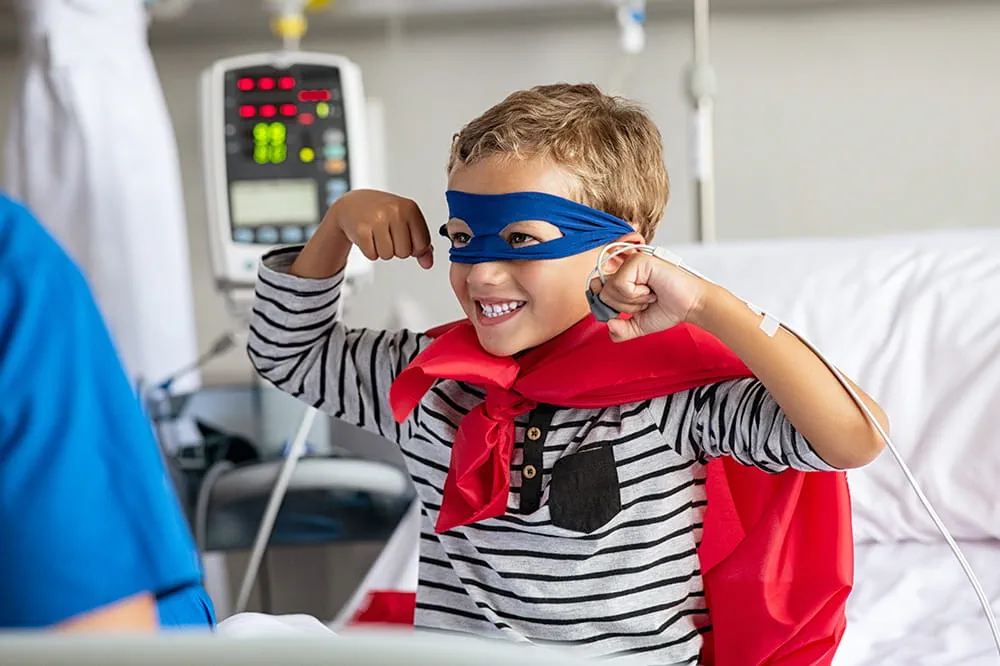
Ranked nationally in pediatric care.
Arkansas Children's provides right-sized care for your child. U.S. News & World Report has ranked Arkansas Children's in seven specialties for 2025-2026.

It's easier than ever to sign up for MyChart.
Sign up online to quickly and easily manage your child's medical information and connect with us whenever you need.
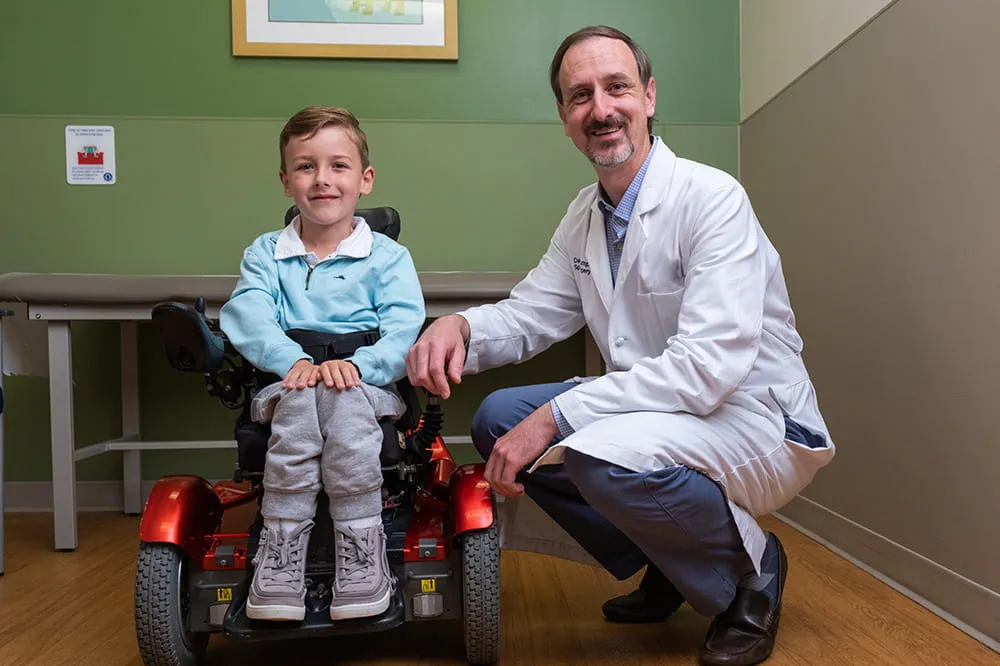
We're focused on improving child health through exceptional patient care, groundbreaking research, continuing education, and outreach and prevention.

When it comes to your child, every emergency is a big deal.
Our ERs are staffed 24/7 with doctors, nurses and staff who know kids best – all trained to deliver right-sized care for your child in a safe environment.

Arkansas Children's provides right-sized care for your child. U.S. News & World Report has ranked Arkansas Children's in seven specialties for 2025-2026.

Looking for resources for your family?
Find health tips, patient stories, and news you can use to champion children.

Support from the comfort of your home.
Our flu resources and education information help parents and families provide effective care at home.

Children are at the center of everything we do.
We are dedicated to caring for children, allowing us to uniquely shape the landscape of pediatric care in Arkansas.

Transforming discovery to care.
Our researchers are driven by their limitless curiosity to discover new and better ways to make these children better today and healthier tomorrow.

We're focused on improving child health through exceptional patient care, groundbreaking research, continuing education, and outreach and prevention.
Then we're looking for you! Work at a place where you can change lives...including your own.

When you give to Arkansas Children's, you help deliver on our promise of a better today and a healthier tomorrow for the children of Arkansas and beyond

Become a volunteer at Arkansas Children's.
The gift of time is one of the most precious gifts you can give. You can make a difference in the life of a sick child.

Join our Grassroots Organization
Support and participate in this advocacy effort on behalf of Arkansas’ youth and our organization.

Learn How We Transform Discovery to Care
Scientific discoveries lead us to new and better ways to care for children.

Learn How We Transform Discovery to Care
Scientific discoveries lead us to new and better ways to care for children.

Learn How We Transform Discovery to Care
Scientific discoveries lead us to new and better ways to care for children.

Learn How We Transform Discovery to Care
Scientific discoveries lead us to new and better ways to care for children.

Learn How We Transform Discovery to Care
Scientific discoveries lead us to new and better ways to care for children.

Learn How We Transform Discovery to Care
Scientific discoveries lead us to new and better ways to care for children.
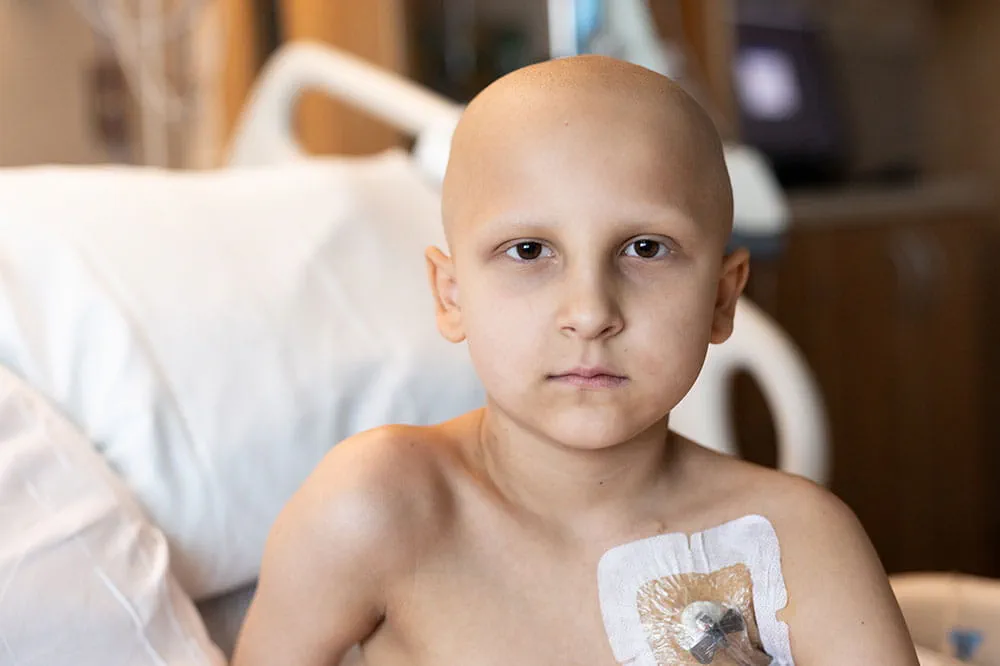
When you give to Arkansas Children’s, you help deliver on our promise of a better today and a healthier tomorrow for the children of Arkansas and beyond.

Your volunteer efforts are very important to Arkansas Children's. Consider additional ways to help our patients and families.

Join one of our volunteer groups.
There are many ways to get involved to champion children statewide.

Make a positive impact on children through philanthropy.
The generosity of our supporters allows Arkansas Children's to deliver on our promise of making children better today and a healthier tomorrow.

Read and watch heart-warming, inspirational stories from the patients of Arkansas Children’s.

Hello.

Arkansas Children's Hospital
General Information 501-364-1100
Arkansas Children's Northwest
General Information 479-725-6800

Arkansas Children's Research Institute (ACRI)
Advances in pediatric medicine begin with a vision for a future in which all children can grow up to be healthy adults. Arkansas Children's Research Institute (ACRI) researchers share this common vision and have devoted their lives to making this vision a reality. At ACRI, children are at the center of everything that we do. Researchers at ACRI are addressing a broad spectrum of children's health concerns, such as the biological mechanisms underlying birth defects; diabetes-related complications; and numerous childhood diseases, including asthma and cancer. Research expertise is diverse, ranging from basic science to clinical and community-based research.
Arkansas Children's Research Institute is a not-for-profit corporation owned by Arkansas Children's, Inc. The Arkansas Children's Hospital Board of Trustees established ACRI in 1989, and ACRI officially opened in May of 1992. The Research Institute Building is located one block south of the main hospital building. ACRI's total research grant base in 1997 was $7.5 million. Today its researchers bring in over $21 million yearly in grants and contracts from federal and private agencies and industry and from philanthropic donations and professional organizations.
Our Mission
At the Arkansas Children's Research Institute (ACRI), we champion children by making them better today and healthier tomorrow.
Our Vision
Our vision is to be on the forefront of innovation in child health and life course research by rapidly transforming discoveries into interventions that define and deliver unprecedented child health.
In support of our vision, we aim to:
- Recruit and retain highly qualified scientists;
- Provide an exceptional environment for research;
- Seek and obtain appropriate funding for research;
- Manage and leverage resources to succeed in our mission;
- Support transfer of discoveries to industry to create jobs in Arkansas;
- Form productive collaborations with other institutions, especially those in Arkansas;
- Promote a strong research capability to support recruitment of the best possible physicians for the campus; and
- Serve as a key learning laboratory for various trainees to learn the disciplines and methodologies of research.

Interested In a Research Study?
We're currently enrolling for a variety of research studies across all ages and demographics.
View Clinical Trials- Ashley Acheson, Ph.D.
- Aline Andres, Ph.D.
- Lauren Appell, M.D.
- Ariel Berlinski, M.D.
- Kevin Bielamowicz, M.D.
- Elisabet Borsheim, Ph.D.
- Andrew W. Brown, PhD, FTOS
- Marie Burdine, Ph.D.
- Thomas Burrow, M.D.
- John Carroll, M.D.
- Jin-Ran Chen, Ph.D.
- Sree V. Chintapalli, Ph.D.
- Divyaswathi Citla Sridhar, M.D.
- Sherry Courtney, M.D.
- Shelley Crary, M.D.
- Melvin Sidney Dassinger, M.D.
- Diana Escalona-Vargas, Ph.D.
- Jason Farrar, M.D.
- Eva Diaz Fuentes, M.D
- Lawrence E. Greiten, MD, MCMSc
- Larry D. Hartzell, MD, FAAP
- Reza Hakkak, Ph.D.
- Trenesha Hill, Ph.D.
- Aaron Kemp, Ph.D.
- Laura James, M.D.
- Stacie Jones, M.D.
- Praveen Juvvadi, Ph.D.
- Colin D. Kay, Ph.D.
- Joshua Kennedy, M.D.
- Mikki Kollisch, M.D., FACS
- Tim Koscik, Ph.D.
- Linda J. Larson-Prior, Ph.D.
- Maya Liza Lopez, M.D.
- Joana M. Mack, M.D.
- Tiffany Miles, Ph.D.
- Thomas Nienaber, MBBS
- Xiawei Ou, Ph.D.
- Tamara Perry, M.D.
- Robert Pesek, M.D.
- Brian Piccolo, Ph.D.
- Ellen van der Plas, Ph.D.
- Craig Porter, Ph.D.
- Konrad Rajab, M.D.
- Gresham Richter, M.D.
- Samrat Roy Choudhury, Ph.D.
- Ronald C. Sanders, Jr., M.D., M.S.
- Michael Schmitz, M.D.
- Amy Scurlock, M.D.
- Megha Sharma, M.D.
- Sarah Sobik, M.D.
- William Steinbach, M.D.
- Graham Strub, M.D., Ph.D.
- Taren M. Swindle, Ph.D.
- Alan Tackett, Ph.D.
- Tomoko Tanaka, MD
- Keshari Thakali, Ph.D.
- Brian Varisco M.D.
- Aravindhan Veerapandiyan, MD
- Marlene Walden, Ph.D.
- Zachary Waldrip, Ph.D.
- Umesh D. Wankhade, PhD
- James Williams, M.D.
- Feliciano Yu, M.D.
If you are unsure of the administrative team member who could best answer your questions, please contact the main office number (501-364-7373), and you will be directed to the appropriate team member.
Research Executive Staff
- Tamara Perry, MD, ACRI President - 501-364-3901
- Phaedra Yount, Vice President of Research Administration and Facilities - 501-364-2469
- Veronica Smith, Vice President of Research Operations - 501-364-6517
- John Fincher, Director of Strategic Operations - 501-364-7368
Research Administrative Staff
- Peggy Bailey, Administrative Assistant - 501-364-7373
- Kelsey Burnham, Grant Development Specialist - 501-364-1303
- April Cooper, Post Award Administration Manger - 501-364-2947
- Keri Davis, Clinical Research Specialist - 501-364-2725
- Andrea Dixon, Corporate Counsel, Research - 501-364-3571
- Brandon Fowlkes, Manager of Animal Care and Husbandry - 501-364-2759
- Julie Fulmer, Informatics Program Manager - 501-364-1860
- Jill Gassaway, Director of Research Integrity - 501-364-2862
- Robyn Graham, Research Account Coordinator - 501-364-6554
- John Gregan, Science Writer/Editor - 501-364-6296
- Gina Hawkins, Pharmacy Coordinator, 501-364-2598
- Dalton Janssen, Clinical Research Programs Manager - 501-364-2391
- Jason Ketcher, PCRU Manager - 501-364-1542
- Teresa McDuffie, Research Accounts Director - 501-364-6546
- Anne McMains, Director of Research Grants and Contracts - 501-364-5143
- Keʹanna Moore, Clinical Research Specialist - 501-364-2721
- Lanette Nimmer, Clinical Research Specialist - 501-364-3577
- Kikumi Ono-Moore, Research Biosafety Specialist - 501-364-2740
- Hannah Parks , Research Pharmacy Coordinator - 501-364-2596
- Stephanie Pate, Executive Assistant - 501-364-6510
- Taylor Peppers, Research Account Coordinator - 501-364-2746
- Robbie Robinette, Senior Software Developer
- Amanda Stapleton, Education Coordinator - 501-364-4008
- Chandra Tanner, Administrative Assistant, 501-364-2700
- Katie Thomason, Executive Assistant - 501-364-6527
- Kimberly Voight, OnCore Administrator - 501-364-5163
- Umesh Wankhade, Director of Animal Research - 501-364-2866
- Aaron Wildschuetz, Facilities and Equipment Manager - 501-364-7519
As part of our efforts to promote academic integrity in science, ACRI has implemented procedures to assure disclosure and appropriate management of all potential conflicts of interest among Academic Staff Members and other research staff operating at ACRI. ACRI's policies and procedures related to financial conflicts of interest assure that the private interests of our researchers and their immediate family members do not improperly influence the design, conduct, or reporting of research. ACRI's researchers are required to disclose financial relationships that can result in a conflict of interest to the ACRI.
Conflicts of interest of ACRI researchers that are related to research funded by the Public Health Service will be made publicly accessible in accordance with 42 C.F.R. Section 50.605.
Learn About Our Summer Science Program.

Summer Science Program
The Summer Science Program at Arkansas Children's Hospital, a program co-sponsored by the UAMS Department of Pediatrics and the Arkansas Children’s Research Institute, gives outstanding sophomore-, junior-, and senior-level college students the experience of a career in academic medicine—in both the clinical and the research aspects.
Learn moreMore Information about ACRI
-
Research
ACRI 3-Year Roadmap
Bookmark ACRI 3-Year RoadmapThis addendum to the Arkansas Children’s Strategic Plan 2020-2025 lays out three thematically oriented goals, each employing the core strategies of the organization’s strategic plan to achieve measurable impact and transform our research enterprise.
-
Research
Resources for Researchers
Bookmark Resources for ResearchersOur staff acts as facilitators to ease the administrative burden of sponsored research. At the same time, we provide an array of unique services, outside the general area of research administration, that have proven to be quite beneficial to the Institute’s cause. -
Research
Participating in Clinical Research
Bookmark Participating in Clinical ResearchThere is a lot to consider before taking part in a research study. These resources will provide general knowledge of what to know or ask to help you make the correct decision regarding participation in clinical research. -
Research
Research Programs and Centers
Bookmark Research Programs and CentersOver 120 pediatric researchers with expertise and experience that span the breadth of medical disciplines comprise ACRI’s roster of investigators who work to fulfill its mission to improve children’s health, development, and well-being through high-quality research.

Support Services
Download the Research Guidebook for detailed descriptions of research support services offered by ACRI Research Administration.
Download
